科研进展
【文献导读】Circulation:托拉塞米与呋塞米对心力衰竭住院患者症状和生活质量的影响:TRANSFORM-HF随机临床试验
托拉塞米与呋塞米对心力衰竭住院患者症状和生活质量的影响:TRANSFORM-HF随机临床试验
文章来源:10.1161/CIRCULATIONAHA.123.064842

编译:杨威,IE-learning翻译团队
Background
Loop diuretics are a primary therapy for the symptomatic treatment of heart failure (HF), but whether torsemide improves patient symptoms and quality of life better than furosemide remains unknown. As pre-specified secondary endpoints, the TRANSFORM-HF trial compared the effect of torsemide versus furosemide on patient-reported outcomes among patients with HF.
背景
袢利尿剂是心力衰竭(HF)对症治疗的主要治疗方法,但托拉塞米是否比速尿更好地改善患者的症状和生活质量仍不清楚。作为预先指定的次要终点,TRANSFORM-HF试验比较了托拉塞米和速尿对心力衰竭患者报告结果的影响。
Methods
TRANSFORM-HF was an open-label, pragmatic, randomized trial of 2,859 patients hospitalized for HF (regardless of ejection fraction) across 60 hospitals in the United States. Patients were randomized in a 1:1 ratio to a loop diuretic strategy of torsemide or furosemide with investigator-selected dosage. This report examined effects on pre-specified secondary endpoints, which included Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) (assessed as adjusted mean difference in change from baseline; range, 0-100 with 100 indicating best health status; clinically important difference, ≥5 points) and Patient Health Questionnaire-2 (PHQ-2) (range, 0-6; score ≥3 supporting evaluation for depression) over 12 months.
方法
TRANSFORM-HF是一项开放、实用的随机试验,纳入了美国60家医院2859例HF住院患者(不论射血分数如何)。患者按1:1的比例随机分配到托拉塞米或呋塞米的循环利尿策略,剂量由研究者选择。该报告检查了对预先指定的次要终点的影响,其中包括堪萨斯城心肌病问卷临床总结评分(KCCQ-CSS)(评估为自基线变化的调整平均差异;范围:0-100,其中100表示最佳健康状态;临床重要差异,≥5分)和患者健康问卷-2 (PHQ-2)(范围0-6;评分≥3分支持抑郁评估),持续12个月。
Results
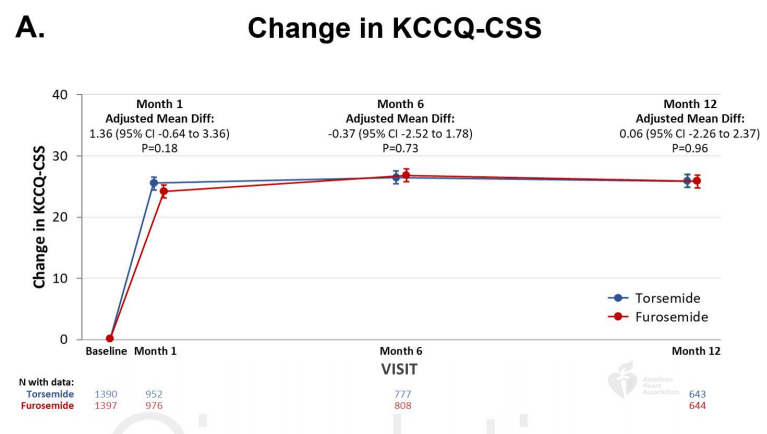
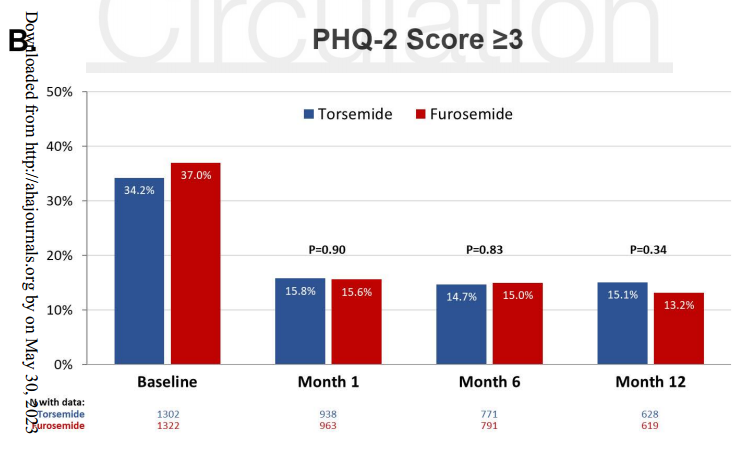
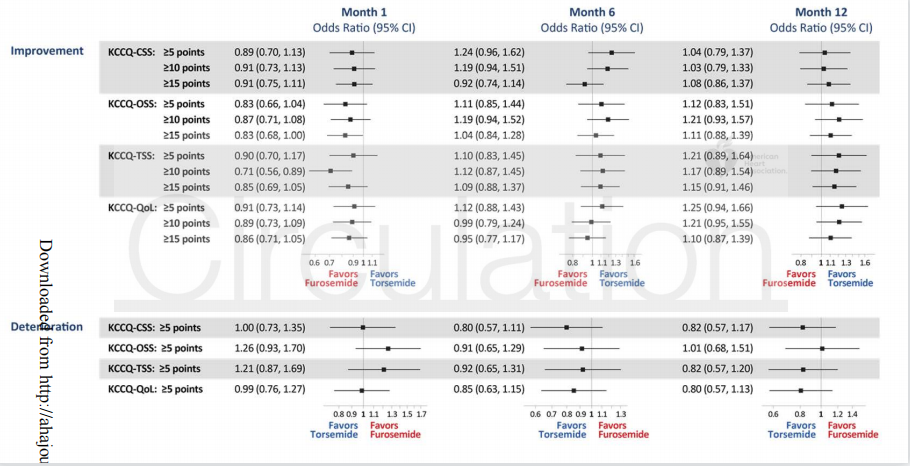
Baseline data were available for 2,787 (97.5%) patients for KCCQ-CSS and 2,624 (91.8%) patients for PHQ-2. Median baseline KCCQ-CSS was 42 (27-60) in the torsemide group and 40 (24-59) in the furosemide group. At 12-months, there was no significant difference between torsemide and furosemide in change from baseline in KCCQ-CSS (adjusted mean difference 0.06 [95% CI, -2.26 to 2.37]; P=0.96) or the proportion of patients with PHQ-2 score ≥3 (15.1% vs 13.2%: P=0.34). Results for KCCQ-CSS were similar at 1-month (adjusted mean difference 1.36 [95% CI, -0.64 to 3.36]; P=0.18) and 6-month follow-up (adjusted mean difference -0.37 [95% CI, -2.52 to 1.78]; P=0.73), and across subgroups by ejection fraction phenotype, New York Heart Association class at randomization, and loop diuretic agent prior to hospitalization. Irrespective of baseline KCCQ-CSS tertile, there was no significant difference between torsemide and furosemide on change in KCCQ-CSS, all-cause mortality, or all-cause hospitalization.
结果
基线数据可获得2787例(97.5%)KCCQ-CSS患者和2624例(91.8%)PHQ-2患者。托拉塞米组的中位基线KCCQ-CSS为42(27-60),呋塞米组为40(24-59)。在12个月时,托拉塞米和呋塞米在KCCQ-CSS(调整后平均差为0.06 [95% CI, -2.26至2.37];P=0.96)或PHQ-2评分≥3的患者比例(15.1% vs 13.2%: P=0.34)中与基线相比的变化无显著差异。KCCQ-CSS的结果在1个月时相似(调整后的平均差为1.36 [95% CI, -0.64至3.36];P=0.18)和6个月随访(调整后平均差异-0.37 [95% CI, -2.52至1.78];P=0.73),并根据射血分数表型、随机分组时纽约心脏协会分级和住院前循环利尿剂进行分组。无论基线KCCQ-CSS分值如何,托拉塞米和呋塞米在KCCQ-CSS、全因死亡率或全因住院方面没有显著差异。
Conclusions
结论
在心力衰竭住院后出院的患者中,与呋塞米相比,托拉塞米的治疗策略在12个月内没有改善症状或生活质量。无论射血分数、既往利尿剂使用情况和基线健康状况如何,托拉塞米和呋塞米对患者报告结果的影响是相似的。
编辑丨国 康
审核丨边 圆 徐 峰
IE-Learning急危重症学习平台
原创声明:文章权限归原作者所有,如需转载请联系我平台。
