科研进展
【文献导读】脓毒症危重患者中美罗培南连续与间歇给药的疗效比较
Continuous vs Intermittent Meropenem Administration in Critically Ill Patients With Sepsis
脓毒症危重患者中
美罗培南连续与间歇给药的疗效比较
The MERCY Randomized Clinical Trial
文章来源:JAMA. doi:10.1001/jama.2023.10598 Published online June 16, 2023
编译:刘海雁,IE-learning翻译团队
IMPORTANCE
Meropenem is a widely prescribed β-lactam antibiotic. Meropenem exhibits maximum pharmacodynamic efficacy when given by continuous infusion to deliver constant drug levels above the minimal inhibitory concentration. Compared with intermittent administration, continuous administration of meropenem may improve clinical outcomes.
重要信息
美罗培南是一种广谱的β-内酰胺类抗菌药。美罗培南在连续输注给药时表现出最大的药效学功效,以使药物水平高于最小抑菌浓度。与间歇给药相比,美罗培南持续给药可能改善临床结局。
OBJECTIVE
To determine whether continuous administration of meropenem reduces a composite of mortality and emergence of pandrug-resistant or extensively drug-resistant bacteria compared with intermittent administration in critically ill patients with sepsis.
目的
确定与间歇给药相比,美罗培南连续给药是否能降低脓毒症危重患者的死亡率和泛耐药或广泛耐药细菌的出现。
DESIGN, SETTING, AND PARTICIPANTS
A double-blind, randomized clinical trial enrolling critically ill patients with sepsis or septic shock who had been prescribed meropenem by their treating clinicians at 31 intensive care units of 26 hospitals in 4 countries (Croatia, Italy, Kazakhstan, and Russia). Patients were enrolled between June 5, 2018, and August 9, 2022, and the final 90-day follow-up was completed in November 2022.
设计、设置和参与者
一项双盲、随机临床试验,入组了4个国家(克罗地亚、意大利、哈萨克斯坦和俄罗斯)26家医院的31个重症监护室的由临床医生开具美罗培南处方治疗的脓毒症或脓毒症休克危重患者。患者于2018年6月5日至2022年8月9日期间入组,最终90天随访于2022年11月完成。
NTERVENTIONS
Patients were randomized to receive an equal dose of the antibiotic meropenem by either continuous administration (n = 303) or intermittent administration (n = 304).
患者随机接受相同剂量的抗生素美罗培南连续给药(n = 303)或间歇给药(n = 304)。

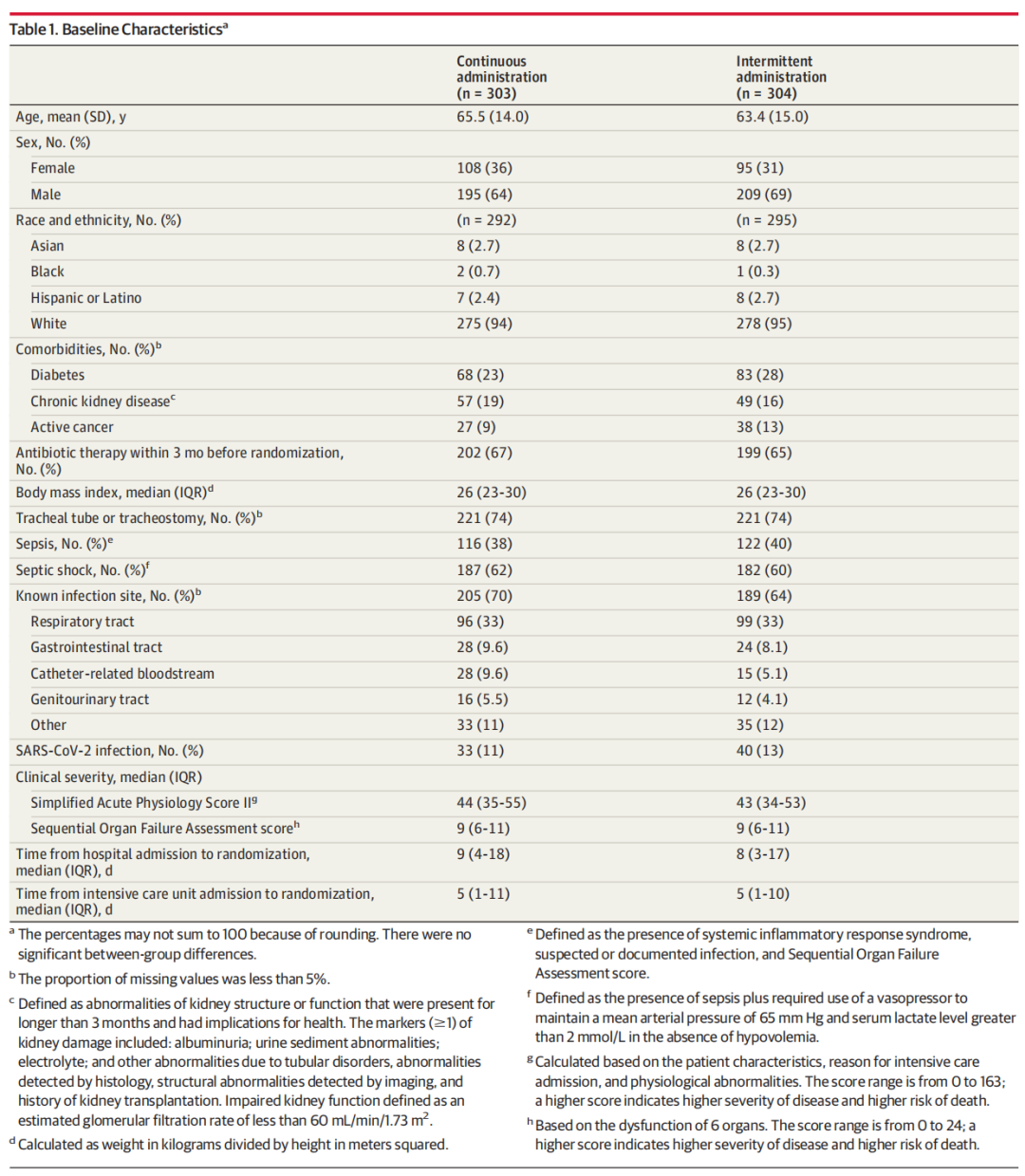
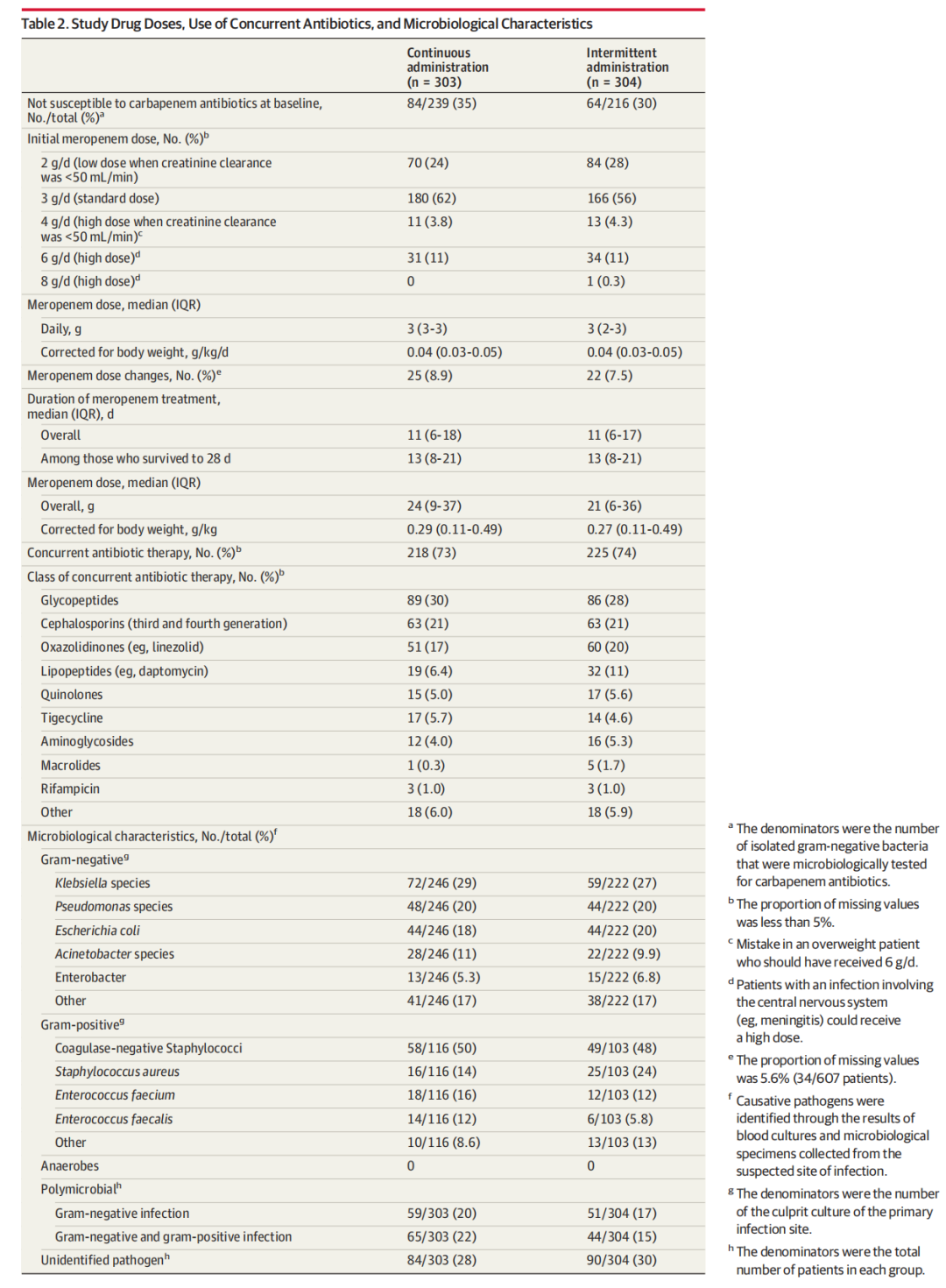
MAIN OUTCOMES AND MEASURES
The primary outcome was a composite of all-cause mortality and emergence of pandrug-resistant or extensively drug-resistant bacteria at day 28. There were 4 secondary outcomes, including days alive and free from antibiotics at day 28, days alive and free from the intensive care unit at day 28, and all-cause mortality at day 90. Seizures, allergic reactions, and mortality were recorded as adverse events.
主要结局和指标
主要结局是全因死亡率和在第28天出现泛耐药或广泛耐药细菌的复合结果。有4个次要结局,包括第28天未使用抗生素的存活天数、第28天未进入重症监护室的存活天数以及第90天的全因死亡率。癫痫发作、过敏反应和死亡被记录为不良事件。
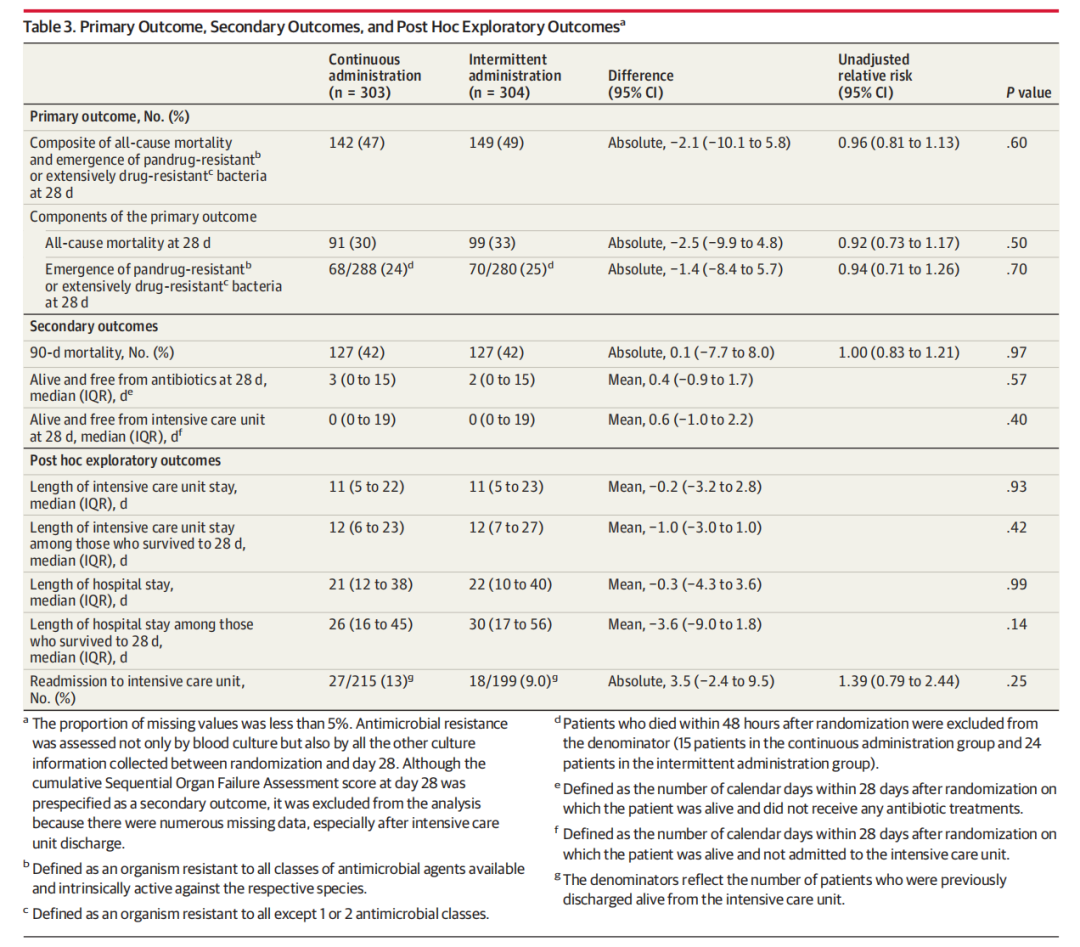
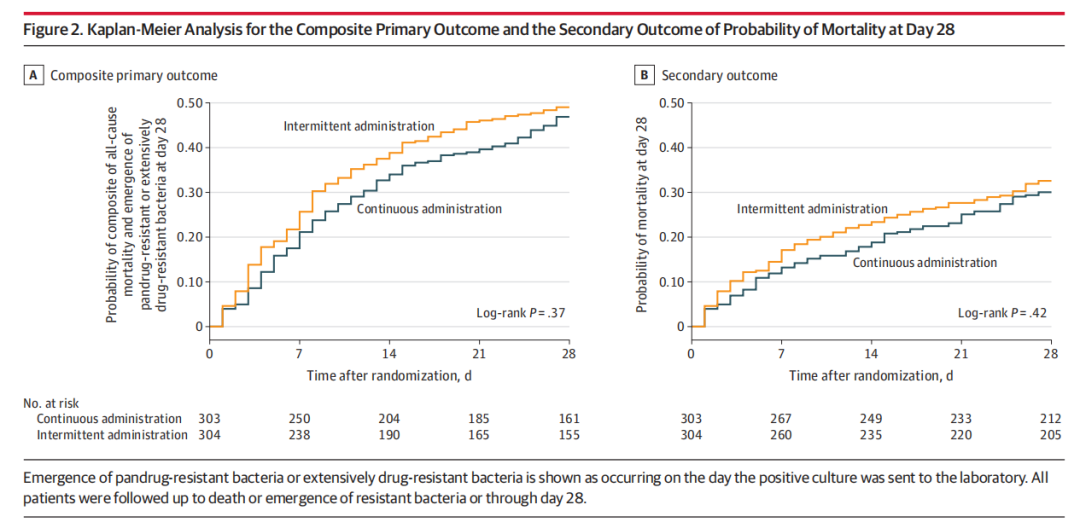
RESULTS
All 607 patients (mean age, 64 [SD, 15] years; 203 were women [33%]) were included in the measurement of the 28-day primary outcome and completed the 90-day mortality follow-up. The majority (369 patients, 61%) had septic shock. The median time from hospital admission to randomization was 9 days (IQR, 3-17 days) and the median duration of meropenem therapy was 11 days (IQR, 6-17 days). Only 1 crossover event was recorded. The primary outcome occurred in 142 patients (47%) in the continuous administration group and in 149 patients (49%) in the intermittent administration group (relative risk, 0.96 [95% CI, 0.81-1.13], P = .60). Of the 4 secondary outcomes, none was statistically significant. No adverse events of seizures or allergic reactions related to the study drug were reported. At 90 days, mortality was 42% both in the continuous administration group (127 of 303 patients) and in the intermittent administration group (127 of 304 patients).
结果
所有607例患者(平均年龄,64 [SD,15]岁; 203例为女性[33%])被纳入28天主要结局的测量中,并完成了90天死亡率随访。大多数(369例患者,61%)发生感染性休克。从入院到随机分组的中位时间为9天(IQR,3-17天),美罗培南治疗的中位持续时间为11天(IQR,6-17天)。仅记录了1起交叉事件。连续给药组142例患者(47%)和间歇给药组149例患者(49%)发生主要结局(相对风险,0.96 [95% CI,0.81-1.13],P = 0.60)。在4个次要结局中,无统计学显著性。未报告与研究药物相关的癫痫发作或过敏反应不良事件。在90天时,连续给药组(303例患者中的127例)和间歇给药组(304例患者中的127例)的死亡率均为42%。
CONCLUSIONS AND RELEVANCE
In critically ill patients with sepsis, compared with intermittent administration, the continuous administration of meropenem did not improve the compositeoutcome of mortality and emergence of pandrug-resistant or extensively drug-resistant bact eria at day 28.
结论和相关性
在脓毒症危重患者中,与间歇给药相比,美罗培南连续给药并未改善第28天死亡率和泛耐药或广泛耐药细菌出现的复合结局。
编辑丨国 康
审核丨杨 威 边 圆 徐 峰
IE-Learning急危重症学习平台
原创声明:文章权限归原作者所有,如需转载请联系我平台。
