科研进展
【文献导读】JAMA Cardiology:在HFmrEF或HFpEF中,达格列净/安慰剂与个体(KCCQ)各指标的关联
Association of Dapagliflozin vs Placebo With Individual Kansas City
Cardiomyopathy Questionnaire Components in Patients With Heart Failure With Mildly Reduced or Preserved Ejection Fraction——A Secondary Analysis of the DELIVER Trial
JAMA Cardiology:在HFmrEF或HFpEF中,达格列净/安慰剂与个体堪萨斯城心肌病问卷(KCCQ)各指标的关联——DELIVER试验的二次分析
文章来源:10.1001/jamacardio.2023.1342
编译:杨秀婷,IE-learning翻译团队
Background
Dapagliflozin has been shown to improve overall health status based on aggregate summary scores of the Kansas City Cardiomyopathy Questionnaire (KCCQ) in patients with heart failure (HF) with mildly reduced or preserved ejection fraction enrolled in the Dapagliflozin Evaluation to Improve the Lives of Patients With Preserved Ejection Fraction Heart Failure (DELIVER) trial. A comprehensive understanding of the responsiveness of individual KCCQ items would allow clinicians to better inform patients on expected changes in daily living with treatment.
达格列净改善HFpEF患者生活的试验(DELIVER)证实 ,根据堪萨斯城心肌病问卷(KCCQ)的汇总评分,达格列净可改善HFmrEF或HFpEF患者的总体健康状况。全面了解个别KCCQ项目的反应性将使临床医生更好地告知患者治疗后日常生活的预期变化。
Methods
To examine the association of dapagliflozin treatment with changes in individual components of the KCCQ. This is a post hoc exploratory analysis of DELIVER, a randomized double-blind placebo-controlled trial conducted at 353 centers in 20 countries from August 2018 to March 2022. KCCQ was administered at randomization and 1, 4, and 8 months. Scores of individual KCCQ components were scaled from 0 to 100. Eligibility criteria included symptomatic HF with left ventricular ejection fraction greater than 40%, elevated natriuretic peptide levels, and evidence of structural heart disease. Data were analyzed from November 2022 to February 2023. Changes in the 23 individual KCCQ components at 8 months.
明确达格列净治疗与KCCQ单个组分变化的相关性。这是对DELIVER试验的事后探索性分析,DELIVER试验是一项随机双盲安慰剂对照试验,于2018年8月至2022年3月在20个国家的353家中心进行。KCCQ评分分别在随机化和1、4和8个月时进行。KCCQ各组成部分的评分汇总从0到100。入选标准包括左心室射血分数大于40%的症状性HF、利钠肽水平升高和结构性心脏病证据。分析了2022年11月至2023年2月的数据。观察8个月时23个单独KCCQ组分的变化。
Results
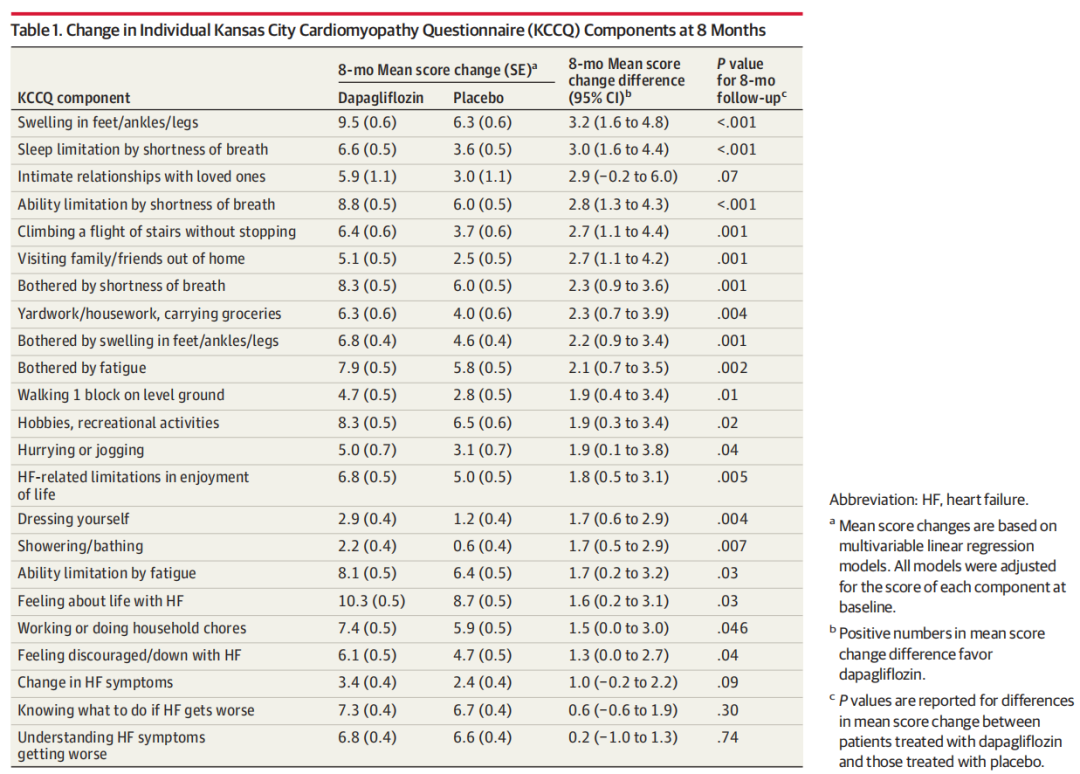
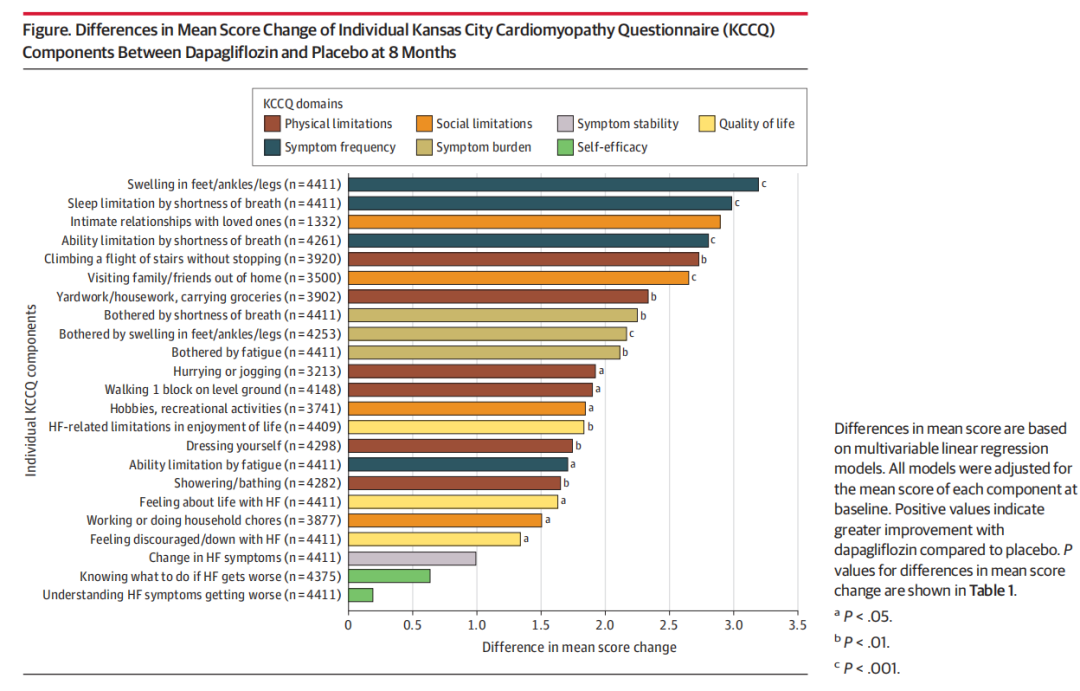
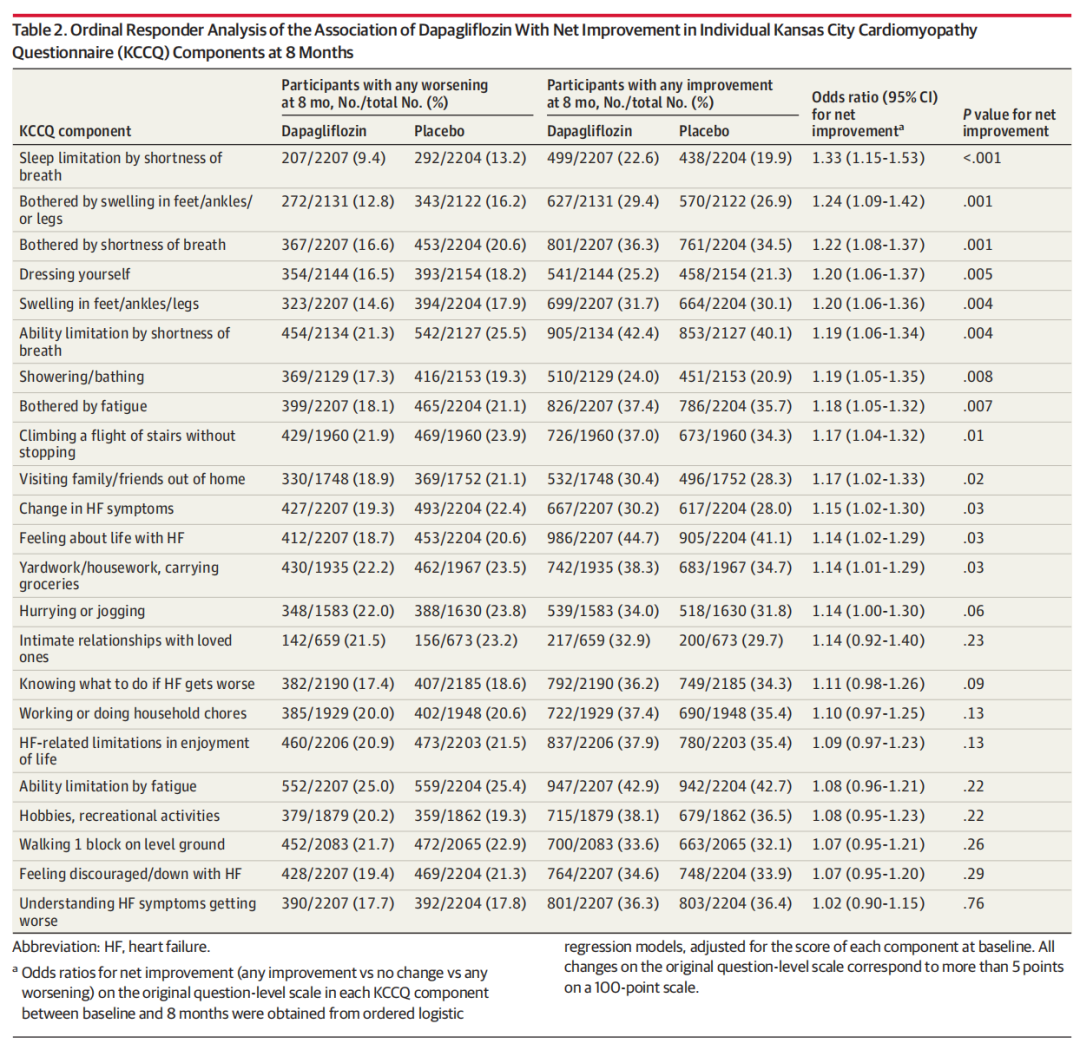
Baseline KCCQ data were available for 5795 of 6263 randomized patients (92.5%) (mean [SD] age, 71.5 [9.5] years; 3344 male [57.7%] and 2451 female [42.3%]). Dapagliflozin was associated with larger improvements in almost all KCCQ components at 8 months compared with placebo. The most significant improvements with dapagliflozin were observed in frequency of lower limb edema (difference, 3.2; 95% CI, 1.6-4.8; P < .001), sleep limitation by shortness of breath (difference, 3.0; 95% CI, 1.6-4.4; P < .001), and limitation in desired activities by shortness of breath (difference, 2.8; 95% CI, 1.3-4.3; P < .001). Similar treatment patterns were observed in longitudinal analyses integrating data from months 1, 4, and 8. Higher proportions of patients treated with dapagliflozin experienced improvements, and fewer had deteriorations across most individual components.
6263例随机化患者中有5795例(92.5%)的基线KCCQ数据可用(平均[SD]年龄:71.5 [9.5]岁; 3344名男性[57.7%]和2451名女性[42.3%])。与安慰剂相比,达格列净与8个月时几乎所有KCCQ组分的较大改善相关。达格列净最显著的改善是下肢水肿的发生率(差异3.2; 95%可信区间为1.6-4.8; P <0.001),呼吸短促导致的睡眠限制(差异,3.0; 95%可信区间为1.6-4.4; P <0.001),以及呼吸短促导致的期望活动受限(差异,2.8; 95%可信区间为1.3-4.3; P < .001)。在整合第1、4和8个月数据的纵向分析中观察到相似的结果。接受达格列净治疗的患者中有更高比例的患者出现改善,并且有更少的患者出现单个组分恶化。
Conclusions
In this study of patients with HF with mildly reduced or preserved ejection fraction, dapagliflozin was associated with improvement in a broad range of individual KCCQ components, with the greatest benefits in domains related to symptom frequency and physical limitations. Potential improvements in specific symptoms and activities of daily living might be more readily recognizable and easily communicated to patients.
结论
在射血分数轻度降低或保留的HF患者的这项研究中,达格列净与广泛的单个KCCQ组分的改善相关,在与症状频率和身体限制相关的领域中获益最大。特定症状和日常生活活动的潜在改善可能更容易识别,并更容易传达给患者。
编辑丨国 康
审核丨边 圆 徐 峰
IE-Learning急危重症学习平台
原创声明:文章权限归原作者所有,如需转载请联系我平台。
