科研进展
【文献导读】NEJM:Olezarsen治疗心血管高危患者的高甘油三酯血症
文章来源:IE-learning IElearning急危重发布时间:2024-06-28 12:02
Olezarsen for Hypertriglyceridemia in Patients at High Cardiovascular Risk
Olezarsen治疗心血管高危患者的高甘油三酯血症
文章来源:DOI: 10.1056/NEJMoa2402309
编译:杨秀婷,IE-learning翻译团队、中国心脏骤停与复苏研究联盟

Reducing the levels of triglycerides and triglyceride-rich lipoproteins remains an unmet clinical need. Olezarsen is an antisense oligonucleotide targeting messenger RNA for apolipoprotein C-III (APOC3), a genetically validated target for triglyceride lowering.
背景降低甘油三酯和富含甘油三酯的脂蛋白的水平仍然是一个尚未满足的临床需求。Olezarsen是一种针对载脂蛋白C-III (APOC3)信使RNA的反义寡核苷酸,而APOC3是经过遗传学验证,可降低甘油三酯的靶点。
In this phase 2b, randomized, controlled trial, we assigned adults either with moderate hypertriglyceridemia (triglyceride level, 150 to 499 mg per deciliter) and elevated cardiovascular risk or with severe hypertriglyceridemia (triglyceride level, ≥500 mg per deciliter) in a 1:1 ratio to either a 50-mg or 80-mg cohort. Patients were then assigned in a 3:1 ratio to receive monthly subcutaneous olezarsen or matching placebo within each cohort. The primary outcome was the percent change in the triglyceride level from baseline to 6 months, reported as the difference between each olezarsen group and placebo. Key secondary outcomes were changes in levels of APOC3, apolipoprotein B, non–high-density lipoprotein (HDL) cholesterol, and low-density lipoprotein (LDL) cholesterol.
在这项2b期随机对照试验中,我们以1:1比例将患中度高甘油三酯血症(甘油三酯水平,150~499 mg/dL)且心血管风险升高的成人或患重度高甘油三酯血症(甘油三酯水平,≥500 mg/dL)的成人分配到50 mg或80 mg队列中。之后在每个队列中以3:1比例分配患者每月接受皮下注射olezarsen或匹配安慰剂。主要结局是甘油三酯水平从基线至6个月的变化百分比,将其报告为每个olezarsen组与安慰剂组的差异。关键次要结局是APOC3、载脂蛋白B、非高密度脂蛋白(HDL)胆固醇和低密度脂蛋白(LDL)胆固醇水平的变化。
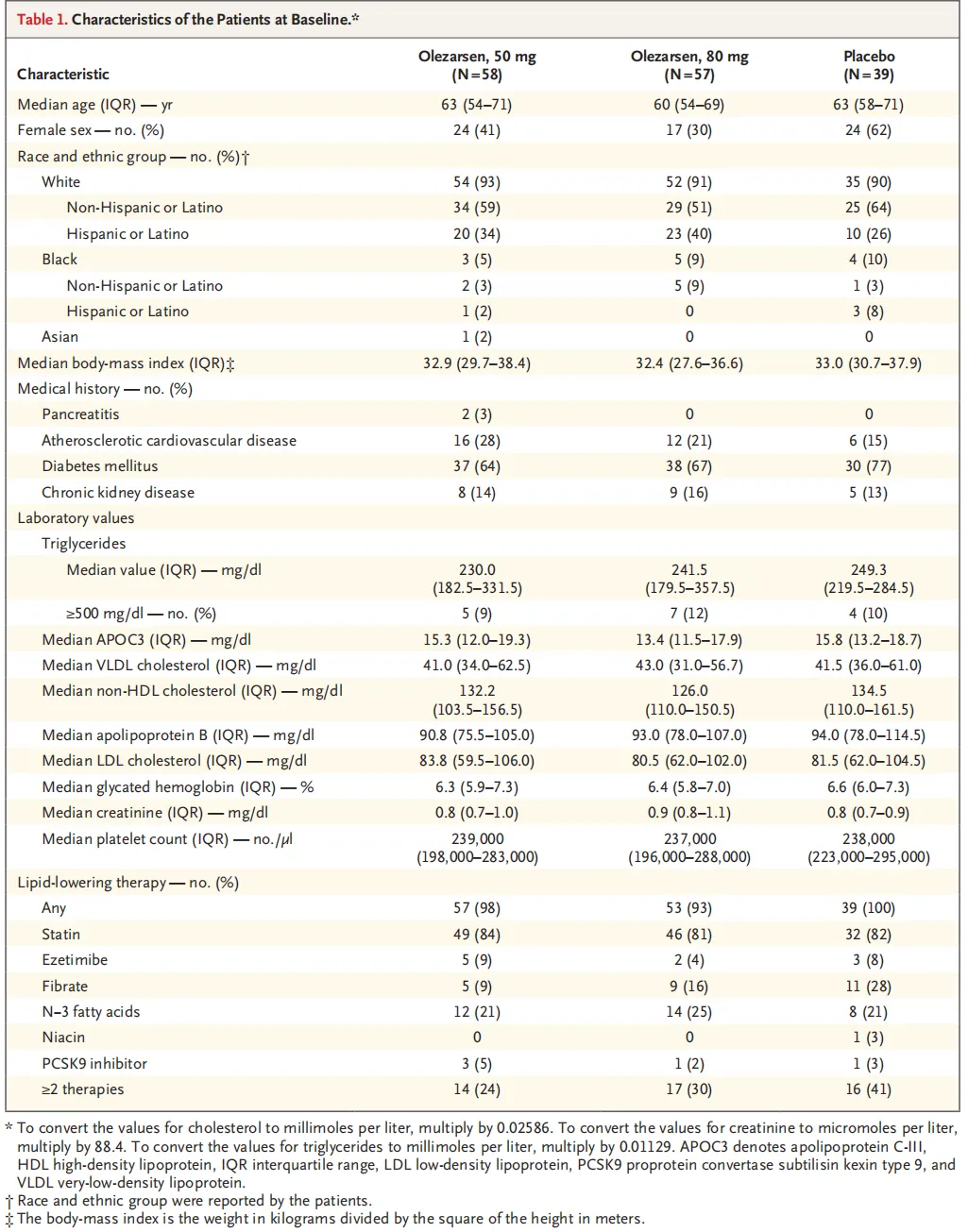
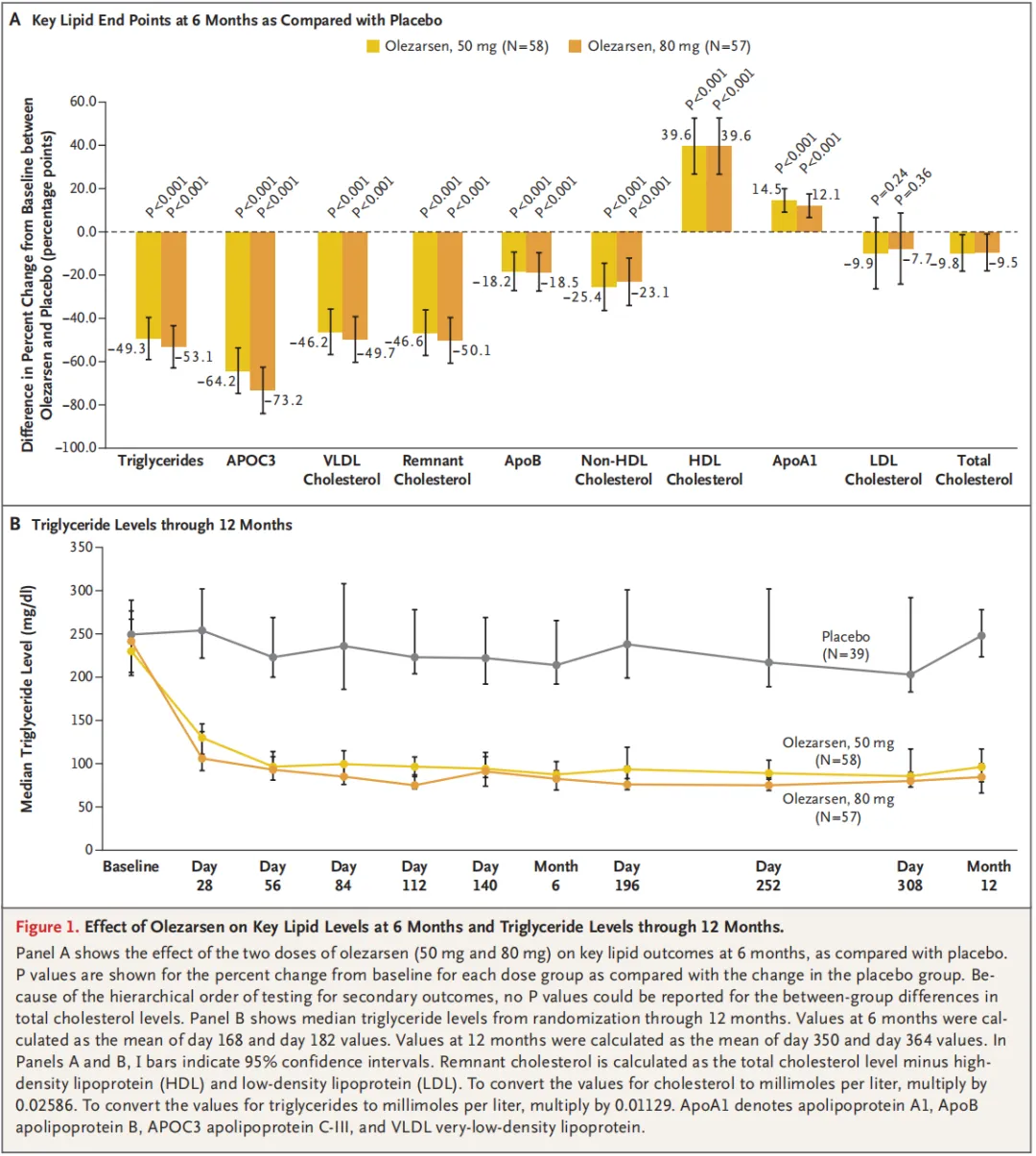
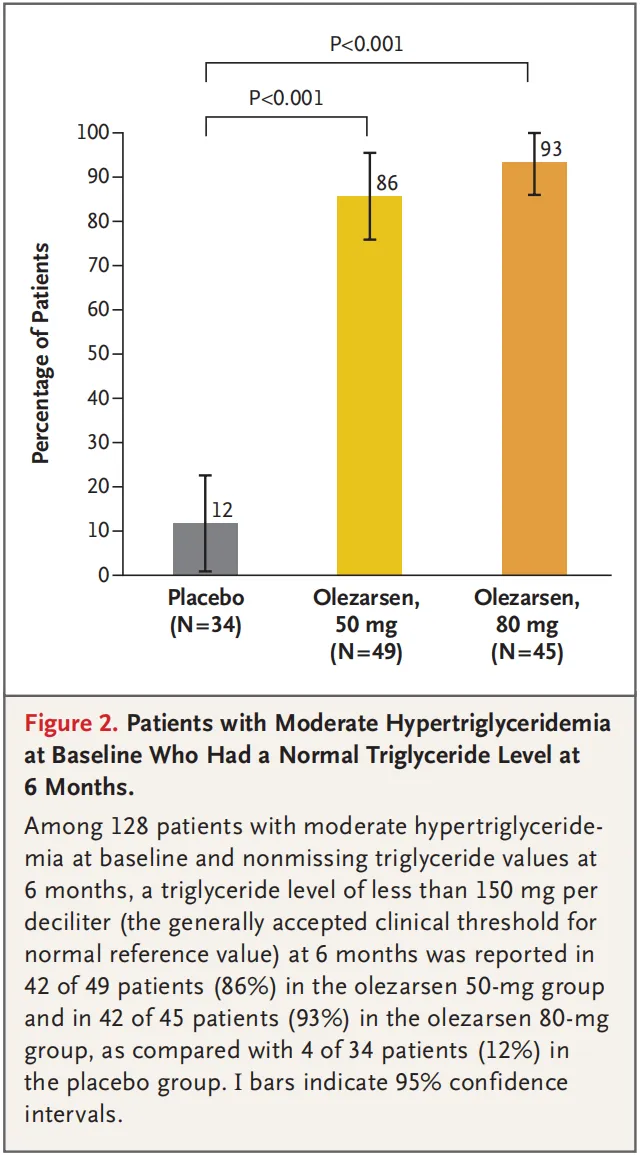
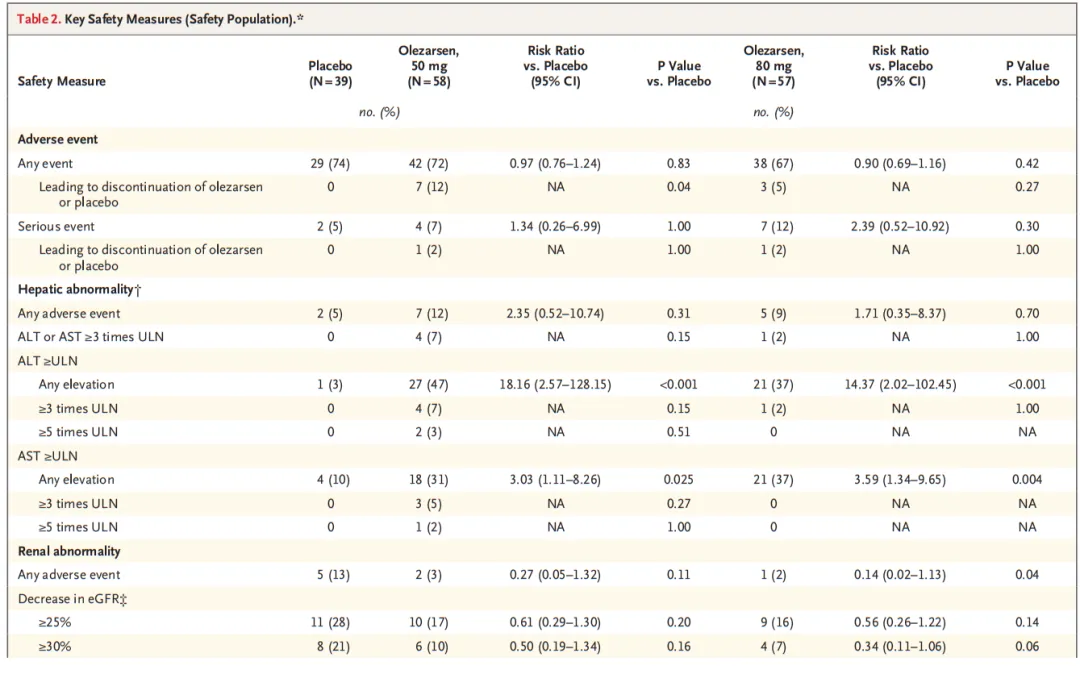
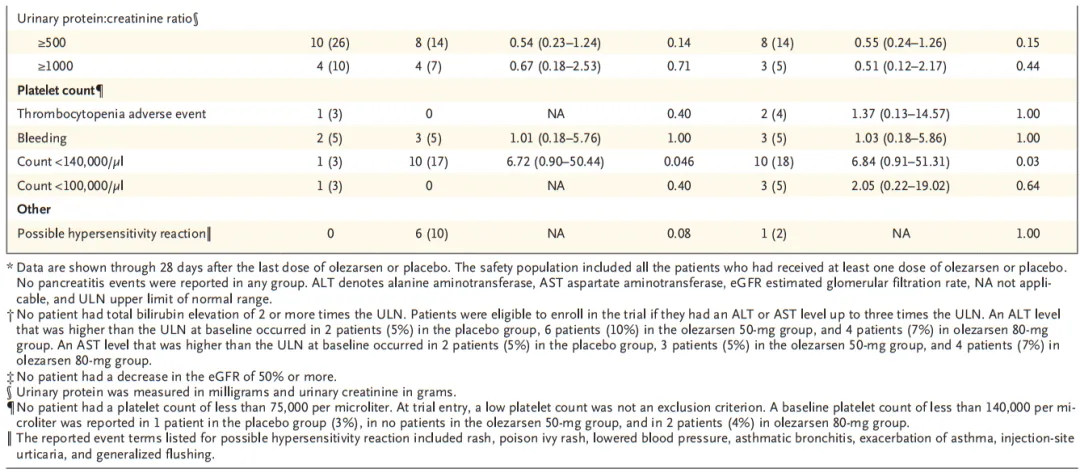
A total of 154 patients underwent randomization at 24 sites in North America. The median age of the patients was 62 years, and the median triglyceride level was 241.5 mg per deciliter. The 50-mg and 80-mg doses of olezarsen reduced triglyceride levels by 49.3 percentage points and 53.1 percentage points, respectively, as compared with placebo (P<0.001 for both comparisons). As compared with placebo, each dose of olezarsen also significantly reduced the levels of APOC3, apolipoprotein B, and non-HDL cholesterol, with no significant change in the LDL cholesterol level. The risks of adverse events and serious adverse events were similar in the three groups. Clinically meaningful hepatic, renal, or platelet abnormalities were uncommon, with similar risks in the three groups.
共计154例患者在北美24个试验中心接受随机分组。患者中位年龄为62岁,中位甘油三酯水平为241.5 mg/dL。与安慰剂相比,50 mg和80 mg剂量olezarsen可将甘油三酯水平分别降低49.3个百分点和53.1个百分点(两项比较的P<0.001)。与安慰剂相比,每种剂量olezarsen还可显著降低APOC3、载脂蛋白B和非HDL胆固醇水平,而LDL胆固醇水平无显著变化。三组发生不良事件和严重不良事件的风险相似。有临床意义的肝、肾或血小板异常并不常见,三组的风险相似。
In patients with predominantly moderate hypertriglyceridemia at elevated cardiovascular risk, olezarsen significantly reduced levels of triglycerides, apolipoprotein B, and non-HDL cholesterol, with no major safety concerns identified.(Funded by Ionis Pharmaceuticals; Bridge–TIMI 73a ClinicalTrials.gov number, NCT05355402.)
在以中度高甘油三酯血症患者为主且心血管风险升高的患者中,olezarsen可显著降低甘油三酯、载脂蛋白B和非HDL胆固醇水平,并且未发现重大安全性问题。(由I Ionis Pharmaceuticals资助;Bridge-TIMI 73a在ClinicalTrials.gov注册号为NCT05355402)。
编辑丨国 康
审核丨杨秀婷 江涛 徐峰
中国心脏骤停与复苏研究联盟
IE-Learning急危重症学习平台
原创声明:文章权限归原作者所有,如需转载请联系我平台。
IE-Learning急危重症学习平台
原创声明:文章权限归原作者所有,如需转载请联系我平台。
